Revised: 20 May 2013
Committees
Minutes of the 144th Medicines Adverse Reactions Committee Meeting - 2 December 2011
INVITED GUESTS AND EXPERTS IN ATTENDANCE
1.2 MINUTES OF THE 143RD MARC MEETING
1.3 DATES OF FUTURE MARC MEETINGS
1.4 POTENTIAL CONFLICTS OF INTEREST
2.1 REPORT ON STANDING AGENDA ITEMS FROM PREVIOUS MEETINGS OF THE MARC
3.1 MATTERS REFERRED TO THE MARC UNDER SECTION 36 OF THE MEDICINES ACT 1981
3.1.1 Consideration of Rosiglitazone-Containing Medicines under Section 36 of the Medicines Act 1981
3.2 MATTERS REFERRED TO THE MARC BY MEDSAFE
4 MATTERS ARISING FROM THE NEW ZEALAND PHARMACOVIGILANCE CENTRE
4.1 CENTRE FOR ADVERSE REACTIONS MONITORING (CARM) QUARTERLY REPORTS
4.1.1 Potential Safety Signals from Single Case Reports
4.1.2 Fatal Cases (Causal Cases Only)
4.1.6 Special Reports: Annual Report of Accident Compensation Commission (ACC) Cases
5.1 CONTINUING MEDICAL EDUCATION
Preface:
In order to protect the privacy of those involved, descriptions of unpublished case reports are not included in these minutes.
Names of individuals have also been deleted where that person's contribution is not in the public domain, or will not shortly be so. For example, the names of those to be approached to write an article are deleted, but the names of those who have contributed to a draft article are not usually deleted. In addition, names are not usually deleted when a contribution has been made in an official capacity.
The material listed as being considered on an issue is not intended to be exhaustive.
The recommendations of the Committee are in bold typeface.
Minutes:
The one hundred and forty-fourth meeting of the Medicines Adverse Reactions Committee (MARC) was held on 2 December 2010 in the Board Room, Medsafe, Wellington, New Zealand. The meeting commenced at 9.30 am and closed at 4 pm.
MARC members present
Associate Professor D Reith (Acting Chair)
Dr L Bryant
Associate Professor M Rademaker
Dr S Sime
Dr M Tatley
Professor P Ellis
Dr S Jayathissa
Dr K Wallis
MARC secretariat present
K Bridgewater (MARC Secretary, Medsafe)
MEDSAFE STAFF IN ATTENDANCE
A Cutfield (Advisor, Pharmacovigilance)
J Hart (Manager, Clinical Risk Management)
C James (Senior Advisor, Pharmacy)
S Kenyon (Senior Advisor, Pharmacovigilance)
Dr E Yousuf (Principal Clinical Advisor)
Invited guests and experts IN ATTENDANCE
Dr R Savage (Senior Medical Assessor, New Zealand Pharmacovigilance Centre)
M Bonne (Senior Policy Analyst, Ministry of Health)
1. Matters of Administration
1.1 Welcome and Apologies
The Acting Chair welcomed the attendees to the meeting. Apologies were received from Associate Professor C Frampton.
Martin Bonne, a Senior Policy Analyst from the Immunisation Team, Ministry of Health, attended as an observer.
1.2 Minutes of the 143rd MARC Meeting
The minutes of the 143rd meeting of the Committee were accepted as a true and accurate record of the meeting.
1.3 Dates of Future MARC Meetings
The dates for the 2011 MARC meetings are scheduled for 10 March 2011, 9 June 2011, 8 September 2011 and 8 December 2011.
1.4 Potential Conflicts of Interest
Committee members submitted their Conflict of Interest Declaration forms to the Secretary. The Chair reminded the MARC members that, in addition to conflicts disclosed in the declaration forms, members should declare conflicts of interest at the commencement of discussion of any relevant agenda item.
There were no potential conflicts of interest which were considered likely to influence the discussions or decisions of the MARC at this meeting.
2. STANDING AGENDA ITEMS
2.1 Report on Standing Agenda Items from Previous Meetings of the MARC
The Committee reviewed the list of outstanding recommendations made by the MARC at previous meetings. Background information on these issues can be found in the minutes of previous MARC meetings from the Medsafe web site at www.medsafe.govt.nz/profs/MARC/Minutes.asp.
Standing agenda items for which the MARC made further recommendations for action are summarised below.
2.1.1 Consideration of Antitussive-expectorant and Antitussive-mucolytic Combination Cough and Cold Medicines under Section 36 of the Medicines Act 1981 - September 2010 minute item 3.2
MARC Recommendation
The Committee recommended that consent be revoked for combination cough and cold medicines containing guaifenesin and dextromethorphan or pholcodine and bromhexine.
The MARC recommended that Medsafe ensure that safety and efficacy data be included in any future applications for antitussive-expectorant or antitussive-mucolytic combination cough and cold medicines.
Outcome
The Minister’s delegate considered the MARC's recommendations and advice provided to him by Medsafe and decided to:
- accep the MARC recommendation that safety and efficacy data be included in any future applications for antitussive-expectorant or antitussive-mucolytic combination cough and cold medicines; and
- not accept the MARC recommendation to revoke the consents for combination cough and cold products containing guaifenesin and dextromethorphan or pholcodine and bromhexine.
However, the Minister’s delegate considers that:
- the use of antitussive-mucolytic and antitussive-expectorant ingredients in a cough and cold preparation appears to be an illogical combination taking into account the individual therapeutic effects of the active ingredients; and
- evidence to identify a patient group that would benefit from such a combination has not been supplied; and
- the use of cough and cold preparations with antitussive-mucolytic and antitussive-expectorant ingredient combinations should therefore be very carefully considered.
Based on the available evidence, the Minister’s delegate considers that any potential risks associated with the use of cough and cold products containing these illogical combinations can be managed by communicating the above key messages to healthcare professionals. Medsafe has been instructed to publish an article in Prescriber Update and liaise with the Pharmaceutical Society of New Zealand to reinforce these messages. In addition, all future advertising and promotional material in relation to Robitussin Cough & Chest Congestion and Duro-Tuss Cough Liquid Expectorant will be expected to be in keeping with the above key messages.
Discussion
The Committee noted that the Minister’s delegate has been advised of the MARC’s recommendations, but has decided to take a wider approach in reaching his final decision. The Committee accepts the process; however, concern was expressed about the effectiveness of the actions planned to manage potential risks associated with these medicines.
It was suggested that the MARC recommendation be followed up with a written letter to the Minister’s delegate expressing disappointment in the decision and requesting that these products be considered by the Medicines Classification Committee (MCC) with a view to their reclassification as Pharmacist-Only medicines. This process was discussed.
Recommendation 1
The MARC recommended writing a letter to the Minister’s delegate acknowledging the decision, expressing disappointment in the decision, and requesting the delegate to consider directing Medsafe to make a Medicines Classification Committee submission for the reclassification of these medicines to Pharmacist-Only.
2.1.2 Review and Proposals for the Adverse Reactions of Current Concern (ARCC) Scheme - June 2010 minute item 3.6; September 2010 minute item 3.6
MARC Recommendation
The Committee recommended that Medsafe and the New Zealand Pharmacovigilance Centre (NZPhVC) proceed with implementing a scheme.
Outcome
Medsafe and the NZPhVC will proceed with implementation and launch.
Discussion
The MARC discussed ways in which the spontaneous reporting scheme could be promoted. The Committee requested to be kept informed about the implementation of the revised ARCC scheme.
Recommendation 2
The MARC recommended that Medsafe provide an update on the scheme to the MARC at a future meeting.
2.1.3 Medsafe Signal Detection and Evaluation Paper - December 2009 minute item 5
MARC Recommendation
In December 2009 the Committee recommended that the Medsafe Signal Detection and Evaluation paper be included in each agenda as an Annex to the Standing Agenda.
Outcome
The Medsafe Signal Detection and Evaluation paper will be included in each agenda as an Annex to the Standing Agenda.
Discussion
The MARC agreed the Medsafe Signal Detection and Evaluation paper provided useful information for the Committee. The Committee considered it would be useful for the paper to include a list of Periodic Safety Update Reports (PSURs) reviewed by Medsafe. The MARC considered the title of the paper should also be changed to better reflect the range of pharmacovigilance activities included in the paper.
Recommendation 3
The MARC recommended that the Medsafe Signal Detection paper include a summary of the PSURs reviewed by Medsafe.
Recommendation 4
The MARC recommended that Medsafe change the name of the Medsafe Signal Detection and Evaluation paper to better reflect the range of pharmacovigilance activities included in the paper.
3. Pharmacovigilance issues
3.1 Matters Referred to the MARC under Section 36 of the Medicines Act 1981
3.1.1 Consideration of Rosiglitazone-Containing Medicines under Section 36 of the Medicines Act 1981
Background
The glitazones were placed on the active monitoring list for annual review in March 2006. In 2007, regulatory action was taken and data sheet updates were made. A Prescriber Update article on fluid retention, heart failure and macular oedema with glitazones was published in November 2007. The results of the annual review are minuted in the March meeting minutes for each subsequent year.
An overview of the data considered at this meeting is attached.
Discussion
The MARC reviewed the report provided by Medsafe.
The Committee was requested to discuss the safety and efficacy of the medicine and whether the information provided was satisfactory to allow continued distribution of rosiglitazone in New Zealand.
The Committee considered that rosiglitazone was effective at reducing hyperglycaemia in diabetic patients. The evidence presented did not indicate superior efficacy compared to other anti-diabetic agents.
The Committee confirmed that both glitazones (rosiglitazone and pioglitazone) are associated with congestive heart failure and debated whether this could contribute to an increased risk of myocardial infarction. It was noted that diabetic patients are already at increased risk of adverse cardiovascular events. The risk of fractures with both glitazones was also reconfirmed. The risk of adverse ischaemic heart events was considered to be the most important factor in reviewing the benefit risk balance for rosiglitazone. The Committee considered that both the meta-analyses and observational data were suggestive of an increased risk of myocardial infarction with rosiglitazone. The published data from the RECORD study indicate a more favourable profile, but these data are being re-analysed at the request of the US Food and Drug Administration.
The Committee agreed that the available data represented an increased risk of myocardial infarction with rosiglitazone treatment which is not apparent with pioglitazone. This additional risk for rosiglitazone was considered to adversely affect the benefit-risk balance and therefore regulatory action was warranted. Overall, the Committee considered the balance of benefits and risks for rosiglitazone was unfavourable. The Committee discussed various regulatory options and considered that as a minimum, further restrictions on use were required. The Committee noted that there were a number of alternative treatments available to patients. Since usage restrictions comparable to those imposed in the US are not applicable in New Zealand the Committee voted 7 to 1 to suspend the consent to distribute rosiglitazone-containing medicines in New Zealand.
The MARC considered that patients and their doctors should be allowed time to initiate alternative treatment; three months was considered to be adequate.
Recommendation 5
The MARC recommended suspension of consents to distribute rosiglitazone containing medicines in New Zealand pending demonstration of a population for whom the balance of benefits and risks is positive.
Recommendation 6
The MARC recommended allowing a period of three months from the Minister or his delegate's acceptance of this recommendation before the suspension commences to allow patients time to transfer to alternative treatments.
3.2 Matters Referred to the MARC by Medsafe
3.2.1 Drospirenone/Ethinyloestradiol: Risk Of Venous Thromboembolism Compared to Other Combined Oral Contraceptives
Background
This topic was first referred to the MARC in September 2008 following local reports of thromboembolic events associated with combined oral contraceptives containing drospirenone - a relatively new and popular oral contraceptive. In subsequent meetings, the Committee considered four recently published studies comparing the risk of venous thromboembolic events (VTE) associated with combined oral contraceptives containing drospirenone with those containing other progestogens, including levonorgestrel (second generation) and desogestrel or gestodene (third generation). Following their review of these studies, the Committee considered firm conclusions could not be drawn from the available data and recommended that the company be asked to undertake a review of the risk of VTE associated with combined oral contraceptives containing drospirenone.
The purpose of the December 2010 report was to provide the Committee with the company’s response to aid in completing the MARC review of this topic.
Discussion
The MARC discussed the report provided by Medsafe which included a review of the company’s analysis of the papers previously seen by the Committee. The Committee noted the strengths and limitations of each study and the additional data provided by the company. The Committee concluded that the risk of VTE for these medicines was comparable to that for ‘second generation’ combined oral contraceptives.
The Committee noted that the company had proposed changes to the data sheets for Yasmin and Yaz. The Committee considered that the proposed wording should be further amended to include VTE incidence rates including those associated with pregnancy as well as reference to the four recent publications. The Committee requested that Medsafe ensure thromboembolic events are listed in the Adverse Effects section of the data sheets.
The Committee proposed that a Prescriber Update article be published to remind prescribers of the risk of VTE with all combined oral contraceptives. The Committee considered that it may be helpful to provide comparative incidence rates in at-risk non-users.
Recommendation 7
The MARC recommended that the data sheets be updated with the wording proposed by the company and the revisions outlined by the Committee.
Recommendation 8
The MARC recommended that an article be published in Prescriber Update as a general reminder of the risk of VTE associated with all combined oral contraceptives.
4. Matters arising from the New Zealand Pharmacovigilance Centre
4.1 Centre for Adverse Reactions Monitoring (CARM) Quarterly Reports
Spontaneous reporting programme
All spontaneous reports presented at the MARC meeting have been assessed by the Centre for Adverse Reactions Monitoring (CARM) and responses have been sent to the reporters. The purpose of these responses is to assist the practitioner to discharge his/her responsibility to patients. These individual replies include as appropriate:
- comment about causality;
- information about similar suspected adverse reactions reported with the same or related medicines;
- prescribing advice;
- advice related to the care of the patient, including information that may assist the practitioner to make a risk:benefit assessment for future treatment; and
- any specific action being taken by the Centre, including entering the reaction into the National Health Index against the patient's name, presenting the case report to the MARC, etc.
Note: In the discussion notes for each report, the case has been given a
causality designation using terms and definitions developed by the World
Health Organization (WHO). The precise definitions are available on the
website of the The Uppsala Monitoring Centre, which is the WHO Collaborating
Centre for International Drug Monitoring -
http://www.who-umc.org/
These designations (certain, probable, possible, unlikely, unclassified
and unclassifiable) refer to the degree of certainty about the relationship
between the medicine and the adverse event. The terms should not be understood
literally. For example, "certain" means that the appropriate elements are
present to match the international definition. It does not mean there is
absolute certainty that the medicine caused the adverse event. Explanations
of these terms can be found on the Medsafe website via the hyperlink at
each causality designation.
4.1.1 Potential Safety Signals from Single Case Reports
4.1.1.1 Hepatic failure and paracetamol (92092)
Discussion
The MARC noted there appears to be some confusion for caregivers about the safety profile and correct dosing of paracetamol in small children.
The Committee agreed that further data should be provided on cases of paracetamol-induced liver injury in children in New Zealand as a result of unintentional overdosing.
Recommendation 9
The MARC recommended that Medsafe and CARM acquire more information on paracetamol-induced liver injury cases in children arising from unintentional overdose in New Zealand and present these data to the MARC at a future meeting.
4.1.2 Fatal Cases (Causal Cases Only)
Members were given a brief description of the fatal reports for which CARM had assessed the causality to be at least possible. The Committee were given the option of requesting that any particular reports be discussed at the current or a subsequent meeting if they considered that there may be a safety issue that prescribers should be informed about or that regulatory action was required.
The Committee did not consider any of the reports required further action.
4.1.3 Special Populations: Serious Cases Associated with Medicines in Children under 18 years (Causal Cases Only)
Reports of serous cases associated with medicines in children under 18 years were briefly outlined for the Committee. The Committee were given the opportunity to request further information on particular reports which may be discussed at a future meeting.
The Committee did not consider any of the reports required further action.
4.1.4 Special Populations: Serious Cases Reporting Adverse Events Following Immunisation Terms with Vaccines in Children under 18 years
Reports of events occurring in children under 18 years were briefly outlined for the Committee. The Committee were given the opportunity to request further information on particular reports which may be discussed at a future meeting.
The Committee did not consider any of the reports required further action.
4.1.5 Special Populations: Serious Non-Fatal Cases Casually Associated with Critical Terms in Patients Over 80 Years
Reports of events occurring in patients over 80 years were briefly outlined for the Committee. The Committee had the opportunity to request further information on particular reports which may be discussed at a future meeting.
The Committee did not consider any of the reports required further action.
4.1.6 Special Reports: Annual Report of Accident Compensation Commission (ACC) Cases
An annual report of ACC case reports received in the period October 2009 to September 2010 was outlined for the Committee. These ACC reports were received by CARM via Medsafe for entry into the CARM database.
Discussion
The Committee noted the Report of ACC case reports. Two case reports with valproate sodium and foetal valproate syndrome (087081and 088850) were noted in relation to a request made by the Neurological Subcommittee of the Pharmacology and Therapeutics Advisory Committee (PTAC) to Medsafe and the MARC to review the risks of valproate use in women of child-bearing age.
The Committee requested further information from Medsafe, including copies of previous MARC reports on this issue as well as international prescribing information to determine whether the current New Zealand datasheet is satisfactory or whether the issue needs to be reviewed again by the MARC.
Recommendation 10
The MARC recommended that Medsafe provide a summary of international prescribing information for comparison with the New Zealand data sheet for valproate. The MARC also requested copies of previous MARC reports and minutes in which the issue of valproate and its risk in pregnancy has been discussed.
5. OTHER BUSINESS
5.1 Continuing Medical Education
[…] gave a presentation on PTAC to the Committee.
6. References
3.1.1 Consideration of Rosiglitazone-Containing Medicines under Section 36 of the Medicines Act 1981
- Review of Benefits and Risks for Rosiglitazone under Section 36 of the Medicines Act 1981.
- GlaxoSmithKline response to the section 36 notice.
- Avandia data sheet. 29 July 2008.
- Graham et al. 2010. Risk of Acute Myocardial Infarction, Stroke, Heart Failure, and Death in Elderly Medicare Patients Treated with Rosiglitazone or Pioglitazone. JAMA doi:10.1001/jama.2010.920
- Brownstein et al. 2010. Rapid Identification of Myocardial Infarction Risk Associated With Diabetes Medications Using Electronic Medical Records. Diabetes Care. 33: 526-531.
- Relevant legislation (Sections 35 and 36 of the Medicines Act 1981).
3.2.1 Drospirenone/Ethinyloestradiol and Risk Of VTE
- Proposed revisions to the data sheets.
- Lidegaard et al. 2009. Hormonal Contraception and risk of venous thromboembolism: national follow-up study. BMJ 339:b2890.
- Van Hylckama Vlieg et al. 2009. The venous thrombotic risk of oral contraceptives, effects of oestrogen dose and progestogen type: results of the MEGA case-control study. BMJ. 339:b2921.
The Acting Chair thanked members and the secretariat for their attendance and closed the meeting at 4 pm.
Associate Professor D Reith
Acting Chair
Medicines Adverse Reactions Committee
ATTACHMENT
Summary of data on the Benefits and Risks for Rosiglitazone
1.0. Background
1.1. Type 2 Diabetes Mellitus
Type 2 diabetes mellitus (T2DM) is a metabolic disorder characterised by high blood glucose in the context of insulin resistance and relative insulin deficiency. Severe complications can result from untreated T2DM including renal failure, blindness, peripheral neuropathy and coronary artery disease. Two thirds of patients with T2DM die from cardiovascular disease.
In 2000 the estimated number of people in New Zealand with diagnosed diabetes was 115,000. This was predicted to rise to over 160,000 in 20 years. The prevalence of diagnosed diabetes is higher among Maori and Pacific people and complications are more common and more severe in these groups. The overall prevalence in the New Zealand population is estimated at 3-4%1.
The absolute risk of coronary artery disease in patients with diabetes is two times higher than in patients without diabetes. Multiple risk factors have been identified to be important in diabetic patients, including control of glucose, blood pressure and lipids as well as smoking status. Additional risk factors include age, ethnicity, duration of diabetes and gender.2
1.2. Thiazolidinediones
Thiazolidinediones (TZD), including rosiglitazone, are agonists at the peroxisomal proliferators activated receptor PPARγ. Glycaemic control is improved by improving insulin sensitivity at sites of insulin resistance: adipose tissue, skeletal muscle and liver.
There is one other medicine in the same class approved for use in treating type 2 diabetes: pioglitazone. In New Zealand only pioglitazone is funded by Pharmac; usage of rosiglitazone is estimated at around 80 patients.
2.0. Efficacy of Rosiglitazone
The efficacy of rosiglitazone was established in the pre-registration clinical trials. The company cites the following publications in support of the efficacy: Charbonnel 19993 , Wolffenbuttel 20004 , Fonseca 20005 and Raskin 20016 .
Long-term, durable anti-hyperglycaemic efficacy of rosiglitazone has been demonstrated in both ADOPT7 and RECORD studies.
In three short-term studies8,9,10 that measured Urinary albumin creatine ratio (ACR), an accepted surrogate marker, there was a reproducible and consistent decrease from baseline in ACR for rosiglitazone treated subjects with T2DM. A decrease from baseline in ACR was also demonstrated in the long term studies ADOPT and RECORD.
The effect of rosiglitazone treatment on microvascular events was assessed in the RECORD11 study There was a trend to a decrease in microvascular events but this was not statistically significant.
Diabetics are at an increased risk of amputations due to peripheral vascular disease, neuropathy and an increased risk of infections. In RECORD there were fewer amputations with rosiglitazone compared to the combination of metformin with sulfonylurea (5 and 15 patients respectively).
Changes in lipids have been evaluated during the clinical development programme for rosiglitazone12,13 . In 26 week, placebo-controlled studies utilising 4mg and 8mg of rosiglitazone the following effects were noted:
- A 14.1% to 18.6% increase in low density lipoprotein-cholesterol (LDL-C) level.
- An 11.4% to 14.2% increase in high density lipoprotein-cholesterol (HDL-C) level.
- No difference in LDL-c/HDL-c ratio at one year.
- Changes in triglycerides have been less consistent and not significantly different from controls.
Across the clinical trial programme consistent reductions in blood pressure, low in magnitude (2-3 mmHg), have been observed with rosiglitazone therapy14,15,16,17 . Pioglitazone has been shown to have a similar effect. Reductions in blood pressure have not been seen with other anti-hyperglycaemic agents.
Dose-related weight gain is associated with TZD use and probably involves a combination of fluid retention and fat accumulation.
In summary rosiglitazone reduces blood glucose levels, has a durability of control and reduces microvascular events, but increases weight and cholesterol levels. The efficacy of rosiglitazone is considered to be acceptable and is comparable to other oral antihyperglycaemics.
Table 1. Summary of Efficacy of Oral Agents from Clinical Trials (provided by GSK)
| Drug Class | Short Term Glycemia | Maximum Length of Trial | Durability of Glycemic Control | Microvascular Events | Macrovascular Benefit |
|---|---|---|---|---|---|
| SU | Up to 1.5% | >10 years | SU>Pbo | Reduced | Not Proven |
| Metformin | Up to 1.5% | >10 years | Met>SU | Probable | Not Proven |
| RSG | Up to 1.5% | 5 years | RSG>Met or SU | Reduced | Not Proven |
| PIO | Up to 1.5% | 3 years | PIO>SU | Probable? | Not Proven |
| DPPIVs | Up to 1.0% | 2-3 years | DPPIV>Pbo | No Data | Unknown |
3.0. Safety of Rosiglitazone
The adverse effects listed in the data sheet are as follows:
| Clinical trial data | |
|---|---|
| General disorders | Oedema |
| Blood and lymphatic system disorders | Anaemia |
| Metabolism and nutrition disorders | Hypercholesterolemia |
| Weight gain | |
| Hypoglycaemia | |
| Cardiac disorders | Congestive heart failure/pulmonary oedema |
| Events typically associated with cardiac ischemia | |
| Musculoskeletal and bone disorders | Bone fractures |
| Postmarketing data | |
| Cardiac disorders | Congestive heart failure/pulmonary oedema |
| Hepatobiliary disorders | Hepatic dysfunction, |
| Skin and subcutaneous tissue disorders | Angioedema |
| Urticaria |
Very rare post-marketing reports of new-onset or worsening diabetic macular
oedema with decreased visual acuity have been reported with rosiglitazone.
4.0. Cardiovascular Risks of Rosiglitazone Treatment
It is the potential for rosiglitazone to cause additional adverse cardiovascular effects such as myocardial infarction is key to the current review of the benefits and risks of rosiglitazone treatment. Therefore the remainder of this document highlights the evidence for an association between rosiglitazone and adverse cardiovascular risks.
4.1. Effect of Rosiglitazone on Surrogates of Cardiovascular Outcomes
Trials on surrogate outcomes have not demonstrated that rosiglitazone is associated with progression of atheroma. Similar studies have shown no progression of atheroma with pioglitazone; there are limited data for other anti-hyperglycaemic agents.
4.2. GSK Integrated Clinical Trial Analyses
This analysis was conducted according to a prospective analysis plan using patient-level data from randomised, double-blind, controlled trials in T2DM patients and analysing major adverse cardiovascular events (MACE) (table 2).
Table 2. MACE (and MACE components): Results from Proportional Hazards Regression Analysis (All Randomised Subjects).
| ICT-52 | Hazard Ratio | Rosiglitazone | Control | |||
|---|---|---|---|---|---|---|
| HR (95%CI) | p-value | Events/ subjects (%) | PY (rate 100PY) | Events/ subjects (%) | PY (rate 100PY) | |
| MACE | 1.121 (0.789-1.593) |
0.525 | 80/10039 (0.80) |
5120.8 (1.56) |
51/6956 (0.73) |
3638.8 (1.40) |
| CV death | 1.264 (0.621-2.572) |
0.518 | 21/10039 (0.21) |
- (0.41) |
12/6956 (0.17) |
- (0.33) |
| MI (SAE) | 1.406 (0.891-2.218) |
0.143 | 55/10039 (0.55) |
- (1.07) |
28/6956 (0.40) |
- (0.77) |
| Myocardial Ischemia | 1.098 (0.890-1.354) |
0.383 | 222/100039 (2.21) |
5120.8 (4.34) |
145/6956 (2.08) |
3638.8 (3.98) |
| Stroke (SAE) | 0.630 (0.333-1.191) |
0.155 | 18/10039 (0.180 |
- (0.35) |
20/6956 (0.29) |
- (0.55) |
Limitations of this analysis were considered by the company to be:
- Most studies were not designed to systematically collect or assess CV endpoints.
- Small numbers of events in the studies.
- Relatively short duration of the studies (< 6 months).
- Not powered to assess cardiovascular outcomes.
Due to these limitations the company considered the results of this analysis to be hypothesis generating. The point estimates were above 1.0 but were not statistically significant.
4.3. FDA Meta-analyses
The US FDA has conducted meta-analyses for both rosiglitazone and pioglitazone (results published on the FDA website). The meta-analysis includes 52 trials. In addition a parallel pioglitazone meta-analysis was conducted in the same manner in order to compare, indirectly, the cardiovascular safety of the TZDs.
Limitations of the meta-analyses considered by the FDA were:
- That most trials were not prospectively designed to evaluate cardiovascular endpoints, however these endpoints endpoints can be reasonably well assessed in clinical trials. Imprecise endpoint assessment would create noise and a bias toward the null.
- Results of trials were known before the statistical analysis plan was developed.
- Statistical significance was not adjusted for multiple testing.
Indirect comparisons between meta-analyses should be considered with caution. However, overall the results suggest that whilst there may be a risk of adverse CV outcomes with rosiglitazone there is no signal for pioglitazone (table 3).
Table 3. Analysis of safety endpoints by meta-analysis
| Meta-analysis | Endpoint | Comparator n (%) | Pioglitazone/ Rosiglitazone n (%) |
Total n (%) | Stratified OR (95% CI) |
|---|---|---|---|---|---|
| Pioglitazone (N=) | 5642 | 6132 | 11774 | ||
| MACE | 63 (1.1) | 54 (0.9) | 117 (1.0) | 0.83 (0.56-1.21) | |
| CV death | 18 (0.3) | 22 (0.4) | 40 (0.3) | 1.18 (0.60-2.34) | |
| MI | 33 (0.6) | 31 (0.5) | 64 (0.5) | 0.91 (0.53-1.53) | |
| Stroke | 16 (0.3) | 10 (0.2) | 26 (0.2) | 0.61(0.24-1.43) | |
| All-cause death | 28 (0.5) | 31 (0.5) | 59 (0.5) | 1.06 (0.61-1.85) | |
| Heart Failure | 50 (0.9) | 75 (1.2) | 125 (1.1) | 1.47 (1.01-2.16) | |
| Rosiglitazone (N=) | 6956 | 10039 | 16995 | ||
| MACE | 39 (0.6) | 70 (0.7) | 109 (0.6) | 1.44 (0.95-2.20) | |
| CV death | 9 (0.1) | 17 (0.2) | 26 (0.2) | 1.46 (0.60-3.77) | |
| MI | 20 (0.3) | 45 (0.4) | 65 (0.4) | 1.80 (1.03-3.25) | |
| Stroke | 16 (0.2) | 18 (0.2) | 34 (0.2) | 0.86 (0.40-1.83) | |
| All-cause death | 17 (0.2) | 29 (0.3) | 46 (0.3) | 1.38 (0.72-2.72) | |
| Heart Failure | 40 (0.6) | 88 (0.9) | 128 (0.8) | 1.93 (1.30-2.93) | |
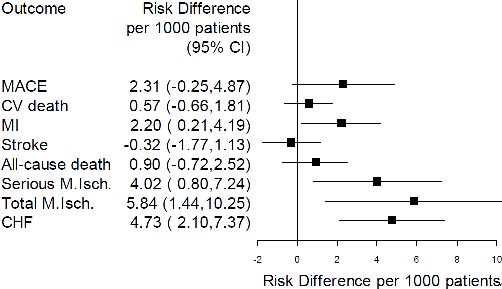
Figure 1. FDA Analysis: Rosiglitazone Sensitivity Analysis, risk difference, all outcomes (on-line slide).
The FDA performed several sensitivity analyses, one of which is illustrated in figure 1. Again the MACE outcome was nearly significant and MI was significantly raised.
The FDA review noted that indirect comparisons between the rosiglitazone and pioglitazone analyses are subject to a number of confounding factors:
Patients in the rosiglitazone meta-analysis were substantially more likely to have received previous treatment for diabetes (78% vs 59%), indicating that there may have been an important difference in prior disease management between the populations studied.
Although average age was similar across studies, a higher proportion of patients in the rosiglitazone studies were aged 65 and over (29% vs 25%).
Patients in the rosiglitazone meta-analysis were predominately from short-term placebo-controlled trials. While the distribution of pioglitazone patients was more uniform across the different trial duration categories.
Proportionately more events occurred in the pioglitazone meta-analysis. The pioglitazone meta-analysis also had more patients in a monotherapy trial group (49% vs 32%) and fewer in the placebo controlled group (39% vs 81%).
The overall event counts in both FDA meta-analyses were low compared to the number of events in the clinical outcomes studies such as RECORD, BARI 2D and PROactive.
Comparing the findings of the meta-analyses with FDA guidance for new antidiabetic drugs shows that rosiglitazone does not meet the requirements. At initial approval the CI upper bound for MACE should be < 1.8, post approval the CI upper bound for MACE OR is ≤1.3. However the FDA view these results as hypothesis generating as the point estimate is below 2.0.
In summary, overall the meta-analyses did not show statistically significant increased risk of major adverse cardiovascular events (MACE); the lower limit was close to 1. However the estimated risk for rosiglitazone was greater relative to comparator. A statistically significant increase in risk of MI was observed that was not seen in the pioglitazone meta-analysis.
4.4. Observational Studies
Observational studies, unlike controlled clinical trials do not have randomised treatment assignation and selection bias may occur. Therefore epidemiologists consider an association with a relative risk of less than 2.0 to be a weak association that could be accounted for by study bias18.
4.4.1 FDA review
The FDA state that they used Cochrane-type systematic review methods to identify relevant studies. Outcomes of interest were: CHF, AMI, stroke, mortality, CAD, HF, angina pectoris, TIA, CVA, CHD, coronary revascularisation, unstable angina, cardiac death, coronary artery reperfusion procedures. Studies eligible for inclusion included:
Endpoints describing cardiovascular risks associated with the use in population settings of rosiglitazone or pioglitazone.
Case-control or cohort design.
Published in a peer-reviewed journal (studies excluded if abstract only).
Twenty-one studies19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39 were identified by the FDA.
Due to differences between the studies, no form of integrated analysis was attempted. The FDA presented the results of their analysis as a series of figures, examples are shown in figures 2 and 3.
The FDA consider that if the risks were truly similar for both drugs one would expect a roughly symmetric distribution of ORs and HRs with similar numbers of point estimates favouring each drug.
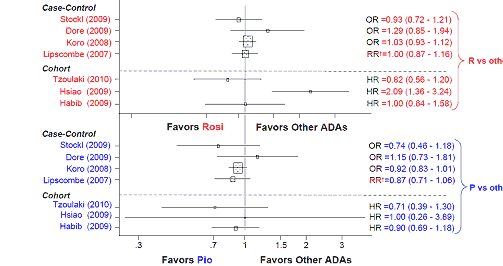
Figure 2. FDA Analysis: Outcome AMI- Rosiglitazone or Pioglitazone versus other Antidiabetic Agents (ADA) (on-line slide).
Comparison of rosiglitazone and pioglitazone with other anti-diabetic agents showed an unfavourable outcome for CHF, as expected. The effect on stoke appeared to be neutral for both. For all cause mortality and acute MI the results for pioglitazone were better than those for rosiglitazone. However, individual point estimates tended to be low and the confidence intervals often spanned 1.0.
In the second analysis rosiglitazone was compared to pioglitazone for cardiovascular outcomes (figures 3).
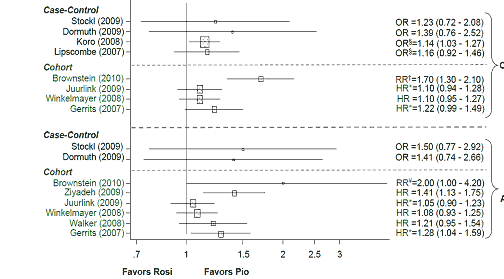
Figure 3. FDA Analysis: Outcome acute myocardial infarction (AMI), rosiglitazone versus pioglitazone (on-line slide).
In general, the results of the comparison between rosiglitazone and pioglitazone favoured pioglitazone.
The FDA state that the strengths of their analysis were:
- Comprehensive and systematic data collection process.
- Methods pre-specified by Cochrane-type protocol.
- Multi-disciplinary FDA team including statistics and epidemiology reviewers.
- Literature searches and adjudication conducted independently by two reviewers.
- Includes studies with direct comparisons of rosiglitazone and pioglitazone, addressing an important knowledge gap.
The FDA state that the limitations were:
- Observational design.
- Individual studies may be subject to various types of bias such as misclassification or confounding by disease severity.
- Function of limitations of individual studies.
- Study level data not patient level.
- Unknown publication bias.
The FDA concluded that overall, comparisons of rosiglitazone and pioglitazone for outcomes including acute myocardial infarction, congestive heart failure and all-cause mortality favour pioglitazone. No studies were identified with results suggesting a protective cardiovascular effect of rosiglitazone compared to pioglitazone. The results are consistent with the results of the FDA’s recently completed meta-analyses of randomised clinical trials.
Potential biases which could affect the interpretation of observational studies of rosiglitazone outlined by the company include:
- Current treatment guidelines only recommend the use of TZDs after metformin and sulfonylureas have been tried. Thus patients using TZDs may have failed other antidiabetic agents and have more co-morbid conditions at baseline and more disease progression.
- Some drug benefit reimbursement organisations restrict reimbursement of rosiglitazone to patients who have failed or have contraindications to other diabetes drugs. Therefore patients taking rosiglitazone are likely to differ from those not taking rosiglitazone in ways that may be associated with cardiovascular outcomes independent of the treatment itself.
- Lack of adjudication of endpoints in observational studies.
- Lack of information on important cardiovascular risk factors e.g. smoking, duration of diabetes, glycaemic control, as well as lack of information on duration of treatment.
- Since 2007, comparison of rosiglitazone and pioglitazone may be biased towards better outcomes for pioglitazone due to over-reporting of ischemic events for rosiglitazone driven by media attention. Furthermore there has been a relative decrease in the use of rosiglitazone in preference for pioglitazone resulting in more new initiators of pioglitazone and confounding any comparison that is made between the two.
- The vast majority of the relative risks estimated are weak associations that are not as high as 2, making it possible that the observed association can be explained by small but systematic bias.
4.4.2. Study by Graham et al40
The retrospective cohort study by Graham et al compared the risks of treatment with rosiglitazone with pioglitazone in Medicare patients in the US. This study was published after the FDA systematic review had been completed. This study attracted some publicity and therefore a brief review is included here. The purpose of the study was to investigate whether rosiglitazone increased the risk of AMI, stroke, heart failure, death or composite outcomes compared with pioglitazone in patients aged 65 years or above.
There were some differences in the baseline characteristics between subjects taking rosiglitazone and those taking pioglitazone, which may have favoured pioglitazone.
The results for the analyses of cardiovascular endpoints tended to show a more favourable outcome for patients taking pioglitazone (table).
Table 4. Graham: Hazard Ratios for Study outcomes comparing rosiglitazone with pioglitazone (on-line slide)
| End point | Unadjusted hazard ratio (95% CI) | Adjusted† hazard ration (95% CI) |
|---|---|---|
| AMI | 1.07 (0.97-1.19) | 1.06 (0.96-1.18) |
| Stroke | 1.31 (1.15-1.49) | 1.27 (1.12-1.45) |
| Heart failure | 1.27 (1.18-1.37) | 1.25 (1.16-1.34) |
| Death | 1.17 (1.07-1.27) | 1.14 (1.05-1.24)‡ |
| AMI or death | 1.13 (1.06-1.21) | 1.11 (1.04-1.19)‡ |
| AMI, stroke, or death | 1.17 (1.10-1.24 | 1.15 (1.08-1.22)‡ |
| AMI, stroke, heart failure, or death | 1.20 (1.14-1.26) | 1.18 (1.12-1.23)‡ |
| † Adjusted for all covariates in AC briefing document;
same model for all end points, ‡ test for PH assumption not met |
||
*Adjustment included variables noted in the baseline characteristics.
The results showed that rosiglitazone was associated with an increased risk of stroke, heart failure and death as well as composite endpoints including AMI or death. The study showed no difference in the risk of AMI between rosiglitazone and pioglitazone.
Major limitations to this study considered by the company include:
- Inadequate duration of follow up time to assess CV outcomes: mean follow up was 4 months.
- The study covered the period of July 2006 to June 2009. During this time frame the entry into the rosiglitazone cohort dropped off substantially, only 22.2% of cohort members commenced treatment with rosiglitazone. Therefore the rosiglitazone cohort consisted mainly of subjects refilling their prescriptions, whereas the pioglitazone cohort consisted of a large proportion of new initiators. This difference biases the results in favour of pioglitazone as this cohort is consistently refreshed and the investigation is of long-term outcomes.
- Demographic imbalances seem to consistently confer a bias favouring pioglitazone.
- The study does not account for the dose-response relationship between rosiglitazone/ pioglitazone and fluid retention and the potential risk for heart failure which could explain the observed difference in CHF between rosiglitazone and pioglitazone.
- The potential for over-diagnosis of CV outcomes in patients treated with rosiglitazone after the publication of the 2007 meta-analysis cannot be ruled out.
- No data were provided on the quality or completeness of medication recording in this database.
- Weak all-cause mortality: HR 1.13 (1.04-1.23). The risk of all-cause mortality was not increased with rosiglitazone in the sensitivity analysis in which patients dying in the hospital after admission or experiencing any study endpoint after stopping the TZD were not counted.
4.4.3. Summary of Results from Observational Studies
Overall the observational studies confirm an increased risk of CHF associated with both TZDs. Studies comparing rosiglitazone with other anti-diabetic agents tend to show a more favourable outcome for the ADAs. Studies comparing rosiglitazone with pioglitazone tend to show a more favourable outcome for pioglitazone. However, it is noted that these are observational studies and are therefore subject to the limitations of these studies.
4.5. RECORD study
Given the limitations of observational studies, definitive assessment of CV outcomes require focus on long-term randomised clinical trials in which confounding factors are controlled.
The Rosiglitazone Evaluated for Cardiac Outcomes and Regulation of Glycaemia in Diabetes (RECORD) study was a post-marketing commitment to the European Medicine Agency EMA.
The RECORD study took place over a 9 year period and was conducted at 364 centres with 1645 events set for adjudication. The company took several steps to attempt to manage issues arising from the open label design. The study protocol was designed in conjunction and approved by European regulatory authorities.
4.5.6. Cardiovascular Outcome Results
The analysis of cardiovascular death and hospitalisation is outlined in table 5. No difference was found between rosiglitazone and the comparator arm. The results confirmed the non-inferiority hypothesis.
A series of pre-specified sensitivity analyses were performed on the primary endpoint. A per-protocol analysis gave similar results to the ITT analysis: HR 1.02 (0.85-1.21). An additional sensitivity analysis excluding some cardiovascular events likely not of atherosclerotic origin gave similar results: HR 0.97 (0.82-1.14).
There were no significant differences between groups for secondary endpoints related to CV events or mortality except for CHF. The incidence of CHF was significantly greater among subjects in the rosiglitazone group. The HR for the secondary composite endpoint of MACE was consistent with the primary endpoint and upper bound of the 95% CI similarly excluded a 20% increase in risk for rosiglitazone compared to metformin/sulfonylurea. While there were more deaths due to CHF in the rosiglitazone group, there was no evidence of an increase in overall risk of CV death.
Table 5. RECORD: Primary and secondary cardiovascular endpoints- overall results
| Rosiglitazone (N=2220) |
Active Control (N=2227) |
HR | Rate diffeence per 1000 person-years | p | |
|---|---|---|---|---|---|
| CV death or CV hospitalisation | 321 | 323 | 0.99 (0.85 to 1.16) | -0.2 (-4.5 to 4.1) | 0.93 |
| All-cause death | 136 | 157 | 0.86 (0.68 to 1.08) | -1.7 (-4.3 to 0.9) | 0.19 |
| CV death | 60 | 71 | 0.84 (0.59 to 1.18) | -0.9 (-2.7 to 0.9) | 0.32 |
| Myocardial infarction* | 64 | 56 | 1.14 (0.80 to 1.63) | 0.6 (-1.1 to 2.4) | 0.47 |
| Stroke* | 46 | 63 | 0.72 (0.49 to 1.06) | -1.4 (-3.1 to 0.2) | 0.10 |
| CV death, MI, or stroke | 154 | 165 | 0.93 (0.74 to 1.15) | -1.0 (-3.9 to 1.9) | 0.50 |
| Heart failure* | 61 | 29 | 2.10 (1.35 to 3.27) | 2.6 (1.1 to 4.1) | 0.0010 |
| Data are numbers, HR (95% CI), or rate differences (95% CI). CV=cardiovascular. MI=myocardial infarction. *Fatal and non-fatal. | |||||
For a safety study, analysis by ITT may not be the optimal analysis. Therefore the FDA did a comparison between ITT and per protocol (PP) populations. No major differences were noted.
Lipid lowering therapy was permitted in RECORD to ensure that patients could be treated to prevailing European guidelines. Simvastatin and atorvastatin were by far the most widely used statins. The mean doses at 5 years were similar for rosiglitazone and control. The proportion of patients with concomitant statin use was higher in the rosiglitazone group beginning in year 1 onwards, reaching a maximum difference of 8.1% of patients by year 5. Consideration was given as to whether this differential could contribute to the difference in atherosclerotic events. Assuming that statins reduce atherosclerotic events by 25% the HR for atherosclerotic events would not be reduced by more than 2% (0.25x0.08). An increase in the HR for the primary endpoint of 2% would not have altered the conclusion of non-inferiority. Despite statin use the lipid levels in rosiglitazone treated subjects remained higher than the comparator groups.
Noted limitations of the RECORD study:
- Unblinded, patients and physicians knew who was taking rosiglitazone.
- Non-inferiority trial, the pre-specified margin was conservative for the time.
- There was asymmetry in addition of 3rd agent/ insulin initiation between groups.
- The composite primary endpoint of cardiovascular death or hospitalisation might contain a wide range of disorders, the hospitalisation could be driven by provider decisions rather than by CV events.
- Adherence to therapy was poor, by the end of the study nearly 50% were no longer taking study drug. The company states this is not relevant as it is exposure that matters in a safety study and exposure was good
- Inappropriate unplanned interim analysis hastily published in 2007 which compromised the integrity of the trial.
- Event rate was very low. Postulated rate was 11%, the observed rate was 2.6%. The annualised MI rate in ACCORD was 1.26%, in RECORD it was 0.49%. The company states that the study had enough power to show non-inferiority at hazard ratios ~1.—or below. The rate was in line with other recent diabetes outcome studies. Sensitivity analyses were all consistent.
- Intent to treat analyses consider those patients as randomised to original study group even through they are taking other drugs, biasing the trial to the null hypothesis. However the sensitivity analysis showed similar results.
- The large excess of statin use in the rosiglitazone arm without reporting of the doses administered may confound the analysis.
- Statin use was equal between arms at the start of the study. Over time use increased more in the rosiglitazone group than in the comparator, the effect on the point estimate was minor.
- The potential for ascertainment bias. Reporting of potential endpoint events was at the discretion of the clinical investigator, who was aware of treatment assignment. There was limited provision to search for events that investigators deemed not to be endpoint events and to upgrade them, therefore biased towards downgrading events. Reanalysis by the FDA of a proportion of subjects resulted in an HR for MACE of 1.07 (0.86-1.33). Further reanalysis for acute MI resulted in a net addition of 19 MIs to the rosiglitazone group and 3 to the control group. The adjusted HR was 1.38 (0.99-1.93; p=NS).
The company believes that despite the challenges of the RECORD study the results are robust and reliable. The results demonstrate that rosiglitazone does not increase the risk of overall cardiovascular morbidity or mortality compared to standard glucose-lowering drugs.
5.0. Outcome of Review by Other Medicines Regulators
5.1. FDA
5.1.1. Summary of decision.
Rosiglitazone may be permitted to remain on the market if the following actions are taken:
- GSK undertakes a restricted access program under a REMS with elements
to assure safe use, including:
- Provision of complete risk information to each patient and documentation in their medical record that the information has been received and understood.
- Documentation from health care providers that each patient receiving
rosiglitazone falls into one of two categories:
- patients currently taking rosiglitazone, or
- patients not already taking rosiglitazone who are unable to achieve glycaemic control on other medications and, in consultation with their health care professional, decide not to take pioglitazone for medical reasons
- Documentation from health care providers that the risk information has been shared with each patient
- Physician, patient, and pharmacist enrollment
- GSK is required to commission an independent re-adjudication of the RECORD study.
- The TIDE trial41 is placed on full clinical hold and the regulatory deadlines for its conduct are rescinded.
5.2. EMA
The European Medicines Agency recommended the suspension of the marketing authorisations for the rosiglitazone-containing anti-diabetes medicines Avandia, Avandamet and Avaglim.
The availability of recent studies has added to the knowledge about rosiglitazone and overall, the accumulated data support an increased cardiovascular risk of rosiglitazone. In view of the restrictions already in place on the use of rosiglitazone, the Committee could not identify additional measures that would reduce the cardiovascular risk. The Committee therefore concluded that the benefits of rosiglitazone no longer outweigh its risks and recommended the suspension of the marketing authorisation of the medicines.
The suspension will remain in place unless the marketing authorisation holder can provide convincing data to identify a group of patients in whom the benefits of the medicines outweigh their risks.
Discussion and Conclusions
The efficacy for rosiglitazone is considered to be acceptable, and is comparable to other anti-diabetic agents. However, rosiglitazone has not been shown to be significantly better than other antidiabetic agents.
A number of adverse reactions to rosiglitazone have been noted. The most important of these are congestive heart failure and bone fractures. These risks have also been noted for pioglitazone. The product information for both TZDs has been updated and restrictions placed on their use which are considered to have sufficiently mitigated these risks.
The current examination is therefore dependent upon whether there is a risk of additional adverse cardiovascular events associated solely with rosiglitazone which changes the benefit risk balance.
The meta-analysis of randomised clinical trials of rosiglitazone performed in 2007 showed an increased risk of myocardial infarction with rosiglitazone treatment. This outcome was not changed by the present re-analysis although the precision of the point estimate increased. However a comparable analysis has now been performed for pioglitazone which does not indicate an increased risk of MACE events. There has been some discussion of these results since strictly speaking the analyses cannot be compared and the point estimates for rosiglitazone are low. Both FDA and the company consider them to be hypothesis generating rather than definitive. However, the results for rosiglitazone do not meet FDA criteria for cardiovascular safety for diabetic medicines. It was also noted that the analyses were able to clearly show the increased risk of CHF with both rosiglitazone and pioglitazone.
The observational studies have many limitations and these have been discussed in this review and by MARC at previous meetings. The quality of these studies was considered to be reasonable, and they were selected using Cochrane principles. In general the results were less favourable for rosiglitazone compared to other anti-diabetic agents including pioglitazone. The results of these studies were consistent with the meta-analysis.
Data from randomised controlled clinical trials was reviewed, the only trial specifically designed to address the current issue was the RECORD study. The published results of this study do not support the results of the meta-analyses or observational studies and are favourable for rosiglitazone. Concerns have been raised regarding this open-label study and have been discussed in this review. These concerns have resulted in the FDA requirement for an independent re-adjudication of the study. It is therefore noted that the current results cannot be considered as reassuring at this point. It is noted that potential biases in the study may result in a less favourable outcome for rosiglitazone post the re-adjudication process.
Considering the difficulties in interpreting this data and the conflicting outcomes for the different study types this issue is referred to the MARC for their expert opinion.
References
- Evidenced Based Best Practice Guideline 2003: Management of Type 2 Diabetes.
- Parchman ML, Zeber JE, Romero RR et al 2007 'Risk of coronary artery disease in type 2 diabetes and the delivery of care consistent with the chronic care model in primary care settings' Med Care; 45: 1129-1134.
- Charbonnel B et al 1999 'Rosiglitazone is superior to glyburide in reducing fasting plasma glucose after 1 year of treatment in type 2 diabetic subjects' Diabetes; 48: A191.
- Wolffenbuttel BHR, Gomist R, Squatrito S et al 2000 'Addition of low dose rosiglitazone to sulfonylurea therapy improves glycemic control in type 2 diabetes subjects' Diabetic Medicine; 17: 40-47.
- Fonseca V, Rosenstock J, Patwardhan R et al 2000 'Effect of metformin and rosiglitazone combination therapy in subjects with type 2 diabetes mellitus' JAMA; 283: 1695-1702.
- Raskin P, Rendell M, Riddle MC et al 2001 'A randomized trial of rosiglitazone therapy in patients with inadequately controlled insulin-treated type 2 diabetes' Diabetes Care; 24: 1226-1232.
- Kahn SE, Haffner SM Heise MA et al 2006 'Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy' NEJM; 355: 2427-2443.
- Bakris G, Viberti G, Weston WM et al 2003 'Rosiglitazone reduces urinary albumin excretion in type II diabetes' J Human Hypertension 17: 7-12.
- Lebovitz HE, Dole JF, Patwardhan R et al 2001 'Rosiglitazone monotherapy is effective in patients with type 2 diabetes' J Clin Endocrinol & Metabolism; 86: 280-288
- Bakris GL, Ruilope LM, McMorn SO et al 2006 'Rosiglitazone reduces microalbuminuria and blood pressure independently of glycaemia in type 2 diabetes patients with microabluminuria' J Hypertension 24: 2047-2055
- Home PD, Pocock SJ, Beck-Nielsen H et al 2009 'Rosiglitazone evaluated for cardiovascular outcomes in oral agent combination therapy for type 2 diabetes (RECORD): a multicentre, randomised, open-label trial' Lancet 373: 2125-2135.
- Malinowski JM, Bolesta S 2000 'Rosiglitazone in the treatment of type 2 diabetes mellitus: a critical review' Clin Ther; 22: 1151-1168.
- Wagstaff AJ, Goa KL 2002 'Rosiglitazone: a review of its use in the management of type 2 diabetes mellitus' Drugs; 62: 1805-1837
- Barnett AH 2003 'Triple oral anti-diabetic therapy in type 2 diabetes' Diabet Med; 20 (suppl 1): 14-16.
- Natali A, Baldeweg S, Toschi E et al 2004 'Vascular effects of improving metabolic control with metformin or rosiglitazone in type 2 diabetes' Diabetes Care; 27: 1349-1357.
- Negro R, Mangieri T, Dazzi D et al 2005 ‘Rosiglitazone effects on blood pressure and metabolic parameters in nondipper diabetic patients’ Diab Res & Clin Pract; 70: 20-25.
- Komajda M, Curtis P, Hanefeld M et al 2008 'Effect of the addition of rosiglitazone to metformin or sulfonylureas versus metformin/sulfonylurea combination therapy on ambulatory blood pressure in people with type 2 diabetes: A randomised controlled trial (the RECORD study)' Cardiovascular Diabetology; 7: 10.doi:10.1186/1475-2840-7-10.
- Shapiro S 2000 'Bias in the evaluation of low-magnitude associations: an empirical perspective' Am J Epidemiol 151: 939-945.
- Azoulay L, Scheider-Lindner V, Dell’aniello S et al 2009 'Thiazolidinediones and the risk of incident strokes in patients with type 2 diabetes: a nested case-control study' Pharmacoepidemiol and Drug Safety 19: 343-350.
- Dore DD, Trivedi AN, Mor V et al 2009 'Association between extent of thiazolidinedione exposure and risk of future myocardial infarction' Pharmacotherapy 29: 775-783.
- Doormuth CR, Maclure M, Carney G et al 2009 'Rosiglitazone and myocardial infarction in patients previously prescribed metformin' PLoS One 4: e6080.
- Stockl KM, Le L, Zhang S et al 2009 'Risk of acute myocardial infarction in patients treated with thiazolidinediones or other antidiabetic medications' Pharmacoepidemiol and Drug Safety 18: 166-174.
- Vanasse A, Carpentier AC, Courteau J et al 2009 'Stroke and cardiovascular morbidity and mortality associated with rosiglitazone use in elderly diabetic patients' Diab Vasc Dis Res 6: 87-93
- Koro CE, Fu Q, Stender M 2008 'An assessment of the effect of thiazolidinedione exposure on the risk of myocardial infarction in type 2 diabetic patients' Pharmacoepidemiol and Drug Safety 17: 989-996.
- Lipscombe LL, Gomes T, Levesque LE 2007 'Thiazolidinediones and cardiovascular outcomes in older patients with diabetes' JAMA 298: 2634-2643.
- Brownstein JS, Murphy SN, Goldmine AB et al 2010 'Rapid identification of myocardial infarction risk associated with diabetes medications using electronic medical records' Diabetes Care 33: 526-531.
- Habit ZA, Togas L, Hasted SL et al 2009 'Relationship between thiazolidinedione use and cardiovascular outcomes and all cause mortality among patients with diabetes: A time updated propensity analysis' Pharmacoepidemiol and Drug Safety 18: 437-447.
- Hsiao FY, Huang WF, Wen YW et al 2009 'Thiazolidinediones and cardiovascular events in patients with type 2 diabetes mellitus: A retrospective cohort study of over 473,000 patients using the national health insurance database in Taiwan' Drug Safety 32: 675-690.
- Juurlink DN, Gomes T, Lipscombe LL et al 2009 'Adverse cardiovascular events during treatment with pioglitazone and rosiglitazone: Population based cohort study' BMJ 339: b2942.
- Shaya FT, Lu Z, Sohn K et al 2009 'Thiazolidinediones and cardiovascular events in high-risk patients with type-2 diabetes mellitus: a comparison with other oral antidiabetic agents.' P.T. 34: 490-501
- Tzoulaki I, Molokhia M, Curcin V et al 2009 'Risk of cardiovascular disease and all cause mortality among patients with type 2 diabetes prescribed oral antidiabetes drugs: Retrospective cohort study using UK general practice research database' BMJ 339: b4731
- Ziyadeh N, McAfee AT, Koro C et al 2009 'The thiazolidinediones rosiglitazone and pioglitazone and the risk of coronary heart disease: A retrospective cohort study using a US health insurance database' Clinical Therapeutics 31: 2665-2677.
- Margolis DJ, Hoffstad, O, Strom BL 2008 'Association between serious ischemic cardiac outcomes ad medications used to treat diabtetes' Pharmacoepidemiol Drug Saf 17: 753-759
- Walker Am, Koro CE, Landon J 'Coronary heart disease outcomes in patients receiving antidiabetic agents in the PharMetrics database 2000-2007' Pharmacoepidemiol and Drug Safety 17: 760-768.
- Winkelmayer WC, Setoguchi S, Levin R et al 2008 'Comparison of cardiovascular outcomes in elderly patients with diabetes who initiated rosiglitazone vs pioglitazone therapy' Arch Intern Med 168: 2368-2375.
- Gerrits CM, Bhattacharya M, Manthena S 2007 'A comparison of pioglitazone and rosiglitazone for hospitalization for acute myocardial infarction in type 2 diabetes.' Pharmacoepidemiol and Drug Safety 16: 1065-1071.
- McAfee AT, Koro C, Landon J et al 'Coronary heart disease outcomes in patients receiving antidiabetic agents' Pharmacoepidemiol Drug Saf 16: 711-25.
- Karter AJ, Ahmed AT, Liu J et al 2005 'Pioglitazone initiation and subsequent hospitalization for congestive heart failure' Diabet Med 22: 986-993
- Rajogopalan R, Rosenson RS, Fernandes AW et al 2004 'Association between congestive heart failure and hospitalization in patients with type 2 diabetes mellitus receiving treatment with insulin or pioglitazone: A retrospective data analysis' Clinical Therapeutics 26: 1400-1410.
- Graham DJ, Quellet-Hellstrom R MaCurdy TE et al 2010 'Risk of acute myocardial infarction, stroke, heart failure and death in Elderly Medicare Patients Treated with Rosiglitazone or Pioglitazone' JAMA 304: 411-418
- A randomised controlled study comparing rosiglitazone with pioglitazone.





