Revised: 27 May 2013
Medicines
Medsafe's performance in the evaluation of new and changed medicines during 2011
From 1 January to 31 December 2011 the following trends and performance were observed.
Work flows
| Application type | Number of applications | |
|---|---|---|
| Received | Consented | |
| Higher risk medicine | 33 (45% abbreviated) | 36 |
| Intermediate risk medicine | 101 (40% abbreviated) | 117 |
| Lower risk medicine | 86 | 90 |
| Changed medicine | 1460 | 1455 |
More medicines are consented than received because the approval process may be longer than 12 months. Applications consented in any given year typically include a proportion lodged in preceding years.
Recorded performance
Number of applications completed within Medsafe's target processing times (in calendar days)
| Application Type/ Event | Initial evaluation | 1st Request for information | Evaluation of 1st response | 2nd Request for information | Evaluation of 2nd response |
|---|---|---|---|---|---|
| Higher risk medicine applications via full evaluation | 12 of 19 (63%) applications completed within 200 days | 92% of applicants responded within 200 days |
95% completed within 200 days |
93% of applicants responded within 120 days | 90% completed within 120 days |
| Higher risk medicine applications via abbreviated evaluation | 20 of 24 applications (83%) completed within 200 days | ||||
| Intermediate risk medicine applications via full evaluation | 54 of 58 applications (93%) completed within 200 days | 92% of applicants responded within 200 days |
97% completed within 120 days |
98% of applicants responded within 120 days |
93% completed within 120 days |
| Intermediate risk medicine applications via abbreviated evaluation | 39 of 46 applications (85%) completed within 100 days | ||||
| Lower risk medicine applications | 91 of 94 applications (97%) completed within 100 days | 87% of applicants responded within 200 days | 93% completed within 120 days |
89% of applicants responded within 120 days | 95% completed within 120 days |
| Application Type/ Event | Initial evaluation | Requests for information | Evaluation responses |
|---|---|---|---|
| Changed medicine notifications | 1435 of 1476 notifications (97%) reviewed within 21 days | Requests for information issued on 20% of notifications
56% of applicants responded within 21 days |
93% reviewed within 21 days |
2% of notifications required referral under s24(5) of the Medicines Act 1981. |
|||
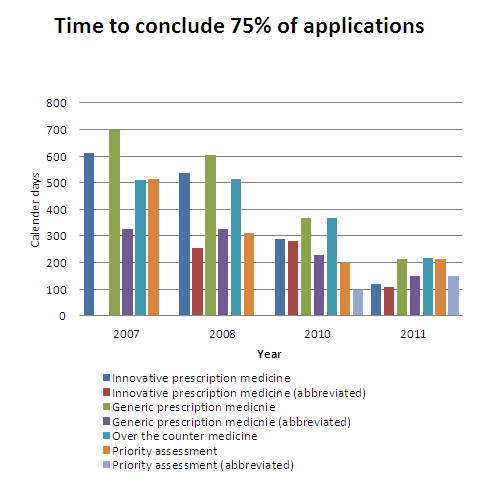
Time to conclude 75% of applications
The time to conclude an application is the total time in calendar days from the day the application was received until an outcome letter is issued. The time includes company response time, which is an average of 100 days for prescription medicine applications and 160 days for over-the counter medicine applications.
| Time in calendar days (75% of applications) |
||||
|---|---|---|---|---|
| Application type | 2007 | 2008 | 2010 | 2011 |
| Innovative prescription medicine | 613 | 535 | 289 | 120 |
| Innovative prescription medicine (abbreviated) | N/A | 253 | 281 | 107 |
| Generic prescription medicine | 697 | 605 | 368 | 212 |
| Generic prescription medicine (abbreviated) | 326 | 326 | 228 | 150 |
| Over the counter medicine | 508 | 512 | 366 | 218 |
| Priority assessment | 515 | 312 | 196 | 212 |
| Priority assessment (abbreviated) | N/A | N/A | 98 | 148 |
Note: some applications received in 2011 are not yet complete. N/A means no applications received of that type.