Revised: 31 May 2013
Publications
Medsafe recalls six products with undeclared prescription medicines
24 September 2010, amended on 14 December 2012: Revised 31 May 2013
Acting Director-General of Health Andrew Bridgman today advised consumers to immediately stop taking six products for erectile dysfunction or the enhancement of sexual performance which contain undeclared prescription medicines, citing significant health risks from their use
The warning, issued under Section 98 of the Medicines Act 1981, followed Medsafe's order for the immediate recall of all batches of the following products - Shaguar, Signature Signergy, “VIGRX” (2 presentations), “VigRx”, and “VigRX Plus”. Photos of the recalled products are available on this page.
Subsequent information provided to Medsafe has confirmed that the “VigRx” and “VigRX Plus” samples seized and tested in 2010 were counterfeits of two natural healthcare products recently approved by Health Canada.
Investigations by Medsafe found that the samples listed below contained one or more undeclared prescription medicines, sildenafil, hydroxyhomosildenafil, thiosildenafil, and/or tadalafil.
“Consumers should immediately stop taking these products and seek medical advice if they have felt unwell when taking any of these products or if they are also taking other medicines,” Mr Bridgman said.
The six products are being promoted and sold in New Zealand by various retailers, including “adult” shops, and over the Internet as products that could enhance sexual performance or treat erectile dysfunction.
The Institute of Environmental Science and Research (ESR) tested samples of the products and found them to contain significant quantities of prescription medicines.
| Product name | Undeclared medicine (dose) | Content |
|---|---|---|
| Shaguar | Tadalafil (20 mg) | Caplet |
| Signature Signergy | Thiosildenafil (63 mg) Hydroxyhomosildenafil (3mg) |
Capsule contents |
| Thiosildenafil (4mcg) | Capsule shell | |
| Counterfeit VIGRX |
Sildenafil (90mg)
Sildenafil (5mg) |
Capsule (green)
Tablet (black) |
| Counterfeit VIGRX |
Sildenafil (65mg)
Sildenafil (2mcg) |
Capsule (white)
Tablet (black) |
| Counterfeit VigRx |
Sildenafil (140 mg) | Tablet |
| Counterfeit VigRX Plus |
Sildenafil (0.3mg)
Tadalafil (0.1mg) |
Caplet |
Tadalafil and sildenafil are the active ingredients of a number of prescription medicines which are used to treat erectile dysfunction. Both tadalafil and sildenafil are known to interfere with some heart medications. Their use can be harmful and even fatal for some people.
Hydroxyhomosildenafil and thiosildenafil are compounds similar in structure to sildenafil. Their safety and efficacy have not been established.
"Since July 2009, Medsafe has identified 45 ostensibly herbal supplements to enhance sexual performance which have been adulterated with similar prescription medicines. The practice of adulterating this type of product with prescription medicines is common," Mr Bridgman cited.
"I advise consumers to treat with extreme caution products purportedly for the treatment of erectile dysfunction or to improve sexual performance offered for sale without a prescription. They should seek medical advice before using them."
Sponsors, distributors, retailers and importers are responsible for the products they sell. They are required under the Medicines Act 1981 to be aware of all the active ingredients contained in their products and to seek approval prior to selling them.
Note: The products referred to in this statement have been found to be adulterated when imported into New Zealand. Other products with the same or similar names may be legally available overseas and / or may have different contents.
ENDS
Questions and Answers
1. What is wrong with these products?
The items have been found to be adulterated with one or more of the prescription medicines tadalafil, sildenafil, hydroxyhomosildenafil and/or thiosildenafil.
Tadalafil and sildenafil are the active ingredients of medicines approved in New Zealand for the management of erectile dysfunction.
Both tadalafil and sildenafil are known to interfere with some heart medications and could be fatal to some individuals. Products containing tadalafil, sildenafil, hydroxyhomosildenafil or other similar substances should only be used on the advice of an authorised New Zealand prescriber after the benefits and risks of use have been assessed.
More information about medicines that interact with tadalafil and sildenafil and other precautions relating to their use can be found by accessing the Consumer Medication Information on the Medsafe website by selecting the “ingredient name” field and typing tadalafil or sildenafil into the search engine at: www.medsafe.govt.nz.
Hydroxyhomosildenafil and thiosildenafil are compounds similar in structure to sildenafil. However, as no products containing either hydroxyhomosildenafil or thiosildenafil have been approved for sale in New Zealand, their safety and efficacy have not been established.
The safety, quality and efficacy of the products in question are unknown as they have not been evaluated prior to their distribution in New Zealand through the medicines approval process.
2. If a consumer is taking one of these products, what should they do?
Consumers are being warned to immediately stop taking these products and seek medical advice from their doctor if they are taking other medicines or have felt unwell when taking the products.
Due to the way they are supplied, there is no reliable information about how many people have taken these products.
Adverse reactions to these products should be reported to the Centre for Adverse Reactions Monitoring: http://nzphvc.otago.ac.nz/report/.
Consumers can also report any concerns to Medsafe: www.medsafe.govt.nz
3. Have these products been removed from sale?
As soon as the results from the ESR testing were received, Medsafe contacted and visited the known distributors and retailers. They were told to remove all stock from their shelves, and quarantine it, as they could not be sure about what these products contained and their sale may be in breach of the medicines legislation. They were also told to notify any other distributors and all mail order customers that they might have sold these products to, advising that the products were being recalled and that all unused products should be returned. Medsafe is continuing to investigate the matter and may take regulatory action if necessary.
4. Have the products on sale in New Zealand been tested?
Medsafe commissioned testing of the four products by ESR. The results of this testing are summarised in the following table.
| Product name | Undeclared medicine (dose) | Content |
|---|---|---|
| Shaguar | Tadalafil (20 mg) | Caplet |
| Signature Signergy | Thiosildenafil (63 mg) and Hydroxyhomosildenafil (3mg) |
Capsule contents |
| Thiosildenafil (4mcg) | Capsule shell | |
| Counterfeit VIGRX |
Sildenafil (90 mg) Sildenafil (5mg) |
Capsule (green) Tablet (black) |
| Counterfeit VIGRX |
Sildenafil (65mg) Sildenafil (2mcg) |
Capsule (white) Tablet (black) |
| Counterfeit VigRx |
Sildenafil (140mg) | Tablet |
| Counterfeit VigRX Plus |
Sildenafil (0.3mg) and Tadalafil (0.1mg) |
Caplet |
Information from the manufacturers and suppliers about the content of erectile dysfunction products (Shaguar and Signature Signergy) sold over-the-counter or over the Internet may be inadequate. It would therefore be prudent not to rely on the labels or other promotional statements made about the ingredients in these products. Further information on VigRX and VigRX Plus can be obtained from the “Licensed natural healthcare products database” on the Health Canada website (www.hc-sc.gc.ca)
5. Where can I find more information about the side effects of tadalafil and sildenafil?
Consumers seeking general information about these active ingredients can access the Consumer Medication Information on the Medsafe website by selecting the “ingredient name” field on the search engine and typing the name of the active ingredient into the search engine at: www.medsafe.govt.nz
6. What about other similar products?
There are many products available from retailers and over the Internet purportedly for the treatment of erectile dysfunction or the improvement of sexual performance. Because of the illicit international trade in some of these products, products that have not been approved for sale in New Zealand may contain potentially harmful substances. Previous investigations by Medsafe have identified a number of products adulterated with prescription medicines used to treat erectile dysfunction, such as tadalafil and sildenafil.
The use of these products can lead to potentially serious consequences, including death. That is why Medsafe has warned against their use.
7. Important advice to traders
Under the medicines legislation, sponsors, distributors, and importers are required to obtain approval before they sell or distribute products intended for a therapeutic purpose.
Section 20 of the Medicines Act 1981 requires medicines to be approved before distribution in New Zealand. A breach of this requirement carries substantial penalties.
On conviction, the maximum penalty for an individual who sells a medicine without first having it approved through the regulatory process administered by Medsafe is $20,000 or up to six months in prison.
On conviction, the maximum penalty for a body corporate which sells a medicine without first having it approved through the regulatory process administered by Medsafe is $100,000.
The Ministry of Health takes breaches of the medicines laws very seriously, especially where patient and consumer safety is put at risk. Regulatory action will be taken as necessary to ensure compliance.
Photos of the recalled products
| Product Name | Undeclared Medicine (Dose) |
Content | Photo |
|---|---|---|---|
| Counterfeit VigRX Plus |
Sildenafil 0.3mg Tadalafil 0.1mg |
Caplet |
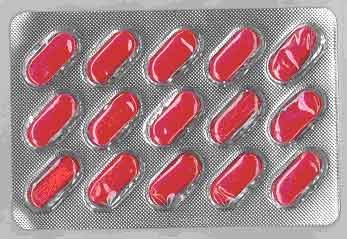 |
| Counterfeit VigRx |
Sildenafil 140mg | Tablet |
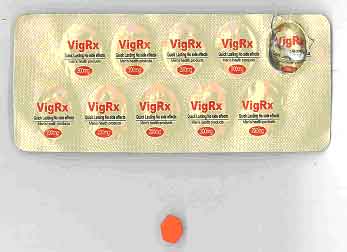 |
| Counterfeit VIGRX |
Sildenafil 90mg Sildenafil 5mcg |
Capsule (Green) Tablet (Black) |
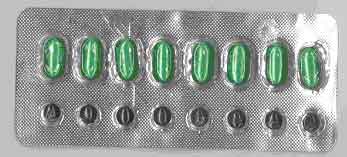 |
| Counterfeit VIGRX |
Sildenafil 65mg Sildenafil 2mcg |
Capsule (White) Tablet (Black) |
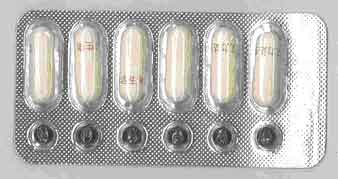 |
| Signature Signergy | Thiosildenafil 0.2mg/mg Hydroxyhomosildenafil 10mcg/mg Thiosildenafil 4mcg |
Capsule Capsule Shell |
 |
| Shaguar | Tadalafil 20mg | Caplet |
 |





