Revised: 2 July 2013
Publications
Statement issued by the Director-General of Health under Section 98 of the Medicines Act 1981: Consumer Warning Regarding Chinese-made Toothpastes that may contain Diethylene Glycol
14 June 2007
Health warning issued under Section 98 of the Medicines Act 1981
Director-General of Health Stephen McKernan is warning consumers against using Chinese-made toothpastes as they may contain diethylene glycol.
In recent weeks the United States Food and Drug Agency announced it had identified a number of Chinese-made toothpastes containing the ingredient diethylene glycol, a toxic material used as a component of antifreeze and some solvents. Consuming diethylene glycol in toothpastes could cause harm in some people, in particular, young children and those with liver or kidney disease.
"As in the United States, the brands of toothpaste that may contain diethylene glycol are not the major brands commonly found in large supermarkets, but are products on sale in smaller discount stores and possibly in stores specialising in Asian products."
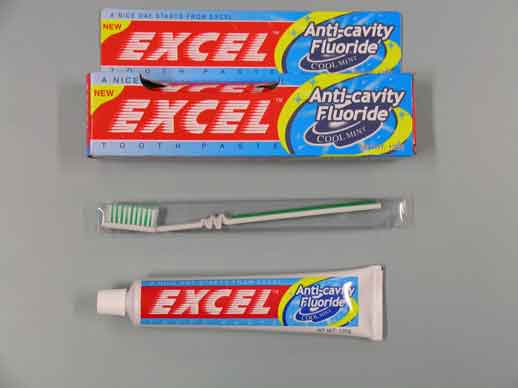
Following information received from a consumer, Medsafe has confirmed the Excel brand of toothpaste identified as containing diethylene glycol has been sold through the South Island Just $2 chain of stores.
The Just $2 chain has withdrawn the toothpaste from its shelves; however, consumers who have purchased this product are advised not to use it. The Just $2 chain will place a recall notice in newspapers inviting consumers to return the product for refund.
Ongoing investigation by Medsafe has identified another 19 companies importing toothpastes from China. Medsafe is urgently contacting these importers for information on whether these toothpastes contain diethylene glycol.
"I advise consumers to check the label on their toothpaste and stop using any products labelled as being made in China or which list diethylene glycol as an ingredient".
While not all Chinese-made toothpastes may contain this ingredient, due to concerns about the potential toxicity of diethylene glycol, a cautious approach is necessary. Further updates will be given following the results of ongoing investigations and testing.
No reports of harm related to the use of toothpastes have been reported in the United States, where the problem was first noted, or in New Zealand.
"While risk of toxicity from the levels of diethylene glycol in these toothpastes is moderately low, it is sufficient to pose a risk of harm to infants and children who eat or swallow toothpaste when cleaning their teeth. In addition, there are potential risks associated with using these toothpastes for individuals with liver or kidney disease".
"If anyone develops symptoms such as nausea, vomiting, headache, lethargy, weakness or confusion after using Chinese-made toothpaste, or otherwise has become unwell with symptoms of kidney or liver disease while using this toothpaste should seek medical advice promptly."
The Director General also advises importers, distributors and retailers of Chinese-made toothpaste that they should check their products for diethylene glycol and contact Medsafe with information about their products.
Photos
Front of Excel pack with toothbrush
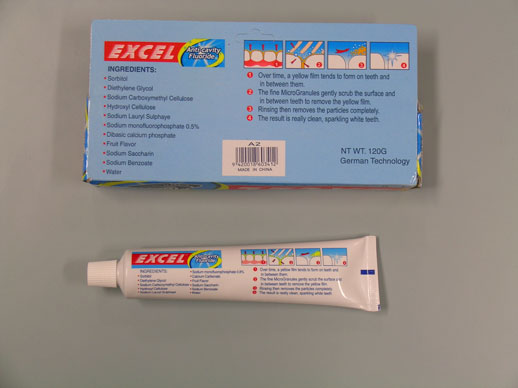
Back of Excel pack
ENDS
For further information please contact:
Anne-Marie Robinson
Media Advisor
Ministry of Health
021 500 634
04 470 0689
Questions and Answers
What symptoms may be associated with consuming Diethylene Glycol ?
Consuming diethylene glycol in toothpastes could cause harm in some people, in particular, young children and those with liver or kidney disease, and its use at high doses in pharmaceuticals has been linked to a number of deaths. Symptoms associated with consuming diethylene glycol may include: nausea, vomiting, headache. Symptoms of moderate exposure include: lethargy, weakness, inebriation, severe gastrointestinal symptoms and low urine output. Severe exposure can lead to liver, blood and kidney disorders, seizure and coma.
Have these products been removed from sale?
Excel toothpaste has been removed from retail sale. Advertisements will appear recalling it from consumers. Other, as yet unidentified products may be on sale. Consumers are advised not to use toothpastes labeled as Made in China or labeled as containing diethylene glycol. Ongoing investigations may lead to further recalls.
If a consumer is taking one of these products what should they do?
Consumers are being warned to immediately stop using these products and seek medical advice from their doctor if they are or have felt unwell when using the products. There is no reliable information about how many people have used these products. Consumers can also report any concerns to Medsafe: www.medsafe.govt.nz
Important advice to importers, distributors and retailers
Products for sale must be of a quality suitable for the purpose and not contain potentially harmful substances. Toothpastes containing fluorides or for which a therapeutic claim is made must go through an assessment by Medsafe before they can be legally marketed in New Zealand.
All importers, distributors and retailers involved in the supply of toothpastes made in China should immediately stop selling the product and check for content of diethylene glycol and/or fluoride. Diethylene glycol may not be listed on the label and its absence from the label may not mean that that it is not an ingredient in the product so information should be sought from the Chinese manufacturer.
Medsafe can be contacted to assist in this process.
Ongoing investigation
Our information so far is that this problem does not affect the big-selling mainstream toothpaste brands.
Approximately 19 companies known to have imported Chinese-made toothpaste during the past 12 months are being contacted to verify the contents of their products.
Information on the toothpaste brands identified in the US as containing diethylene glycol can be obtained from www.fda.gov/oc/opacom/hottopics/toothpaste/faqs.html.
Toothpastes identified by USFDA as containing diethylene glycol include:
- Cooldent Fluoride
- Cooldent Spearmint
- Cooldent ICE
- Dr. Cool
- Everfresh Toothpaste
- Superdent Toothpaste
- Clean Rite Toothpaste
- Oralmax Extreme
- Oral Bright Fresh Spearmint Flavor
- Bright Max Peppermint Flavor
- ShiR Fresh Mint Fluoride Paste
- DentaPro
- DentaKleen
- DentaKleen Junior





